Prostate cancer is the third most common type of cancer in the United States, after breast cancer and lung cancer. In 2018 alone, 164,690 individuals were diagnosed with prostate cancer, accounting for nearly 10% of all new cancer cases, and 29,430 deaths were attributed to the disease.1 Prostate cancer is most frequently diagnosed in men aged 65 to 74 years (median age, 66 years). More than 98% of patients with prostate cancer survive ≥5 years; however, the 5-year survival rate drops to 30% for patients with metastatic disease.1
In 2017, the treatment of prostate cancer accounted for approximately $14.8 billion, representing a $3 billion increase since 2010 ($11.8 billion).2
The treatment of prostate cancer is based on the disease stage, aggressiveness, extent of the spread, the patient’s overall health status, and the benefits and risks of specific treatments.3 Treatment approaches include active surveillance, radical prostatectomy, radiation therapy, hormone therapy (eg, luteinizing hormonereleasing hormones, anti-androgen drugs, and orchiectomy), chemotherapy, biologic therapy (immunotherapy), and cryoablation.3
If prostate cancer metastasizes, androgen-deprivation therapy is typically continued to reduce the patient’s androgen levels; however, in the majority of patients, the disease eventually progresses to castration-resistant prostate cancer (CRPC) despite reduced (ie, castrate) levels of serum testosterone (<50 ng/dL), as defined by the Prostate Cancer Clinical Trials Working Group.4-6
FDA Approves Yonsa, a New Abiraterone Acetate Formulation
On May 23, 2018, the US Food and Drug Administration (FDA) approved a fine-particle formulation of abiraterone acetate (Yonsa; Sun Pharma), an oral cytochrome (CY) P17 inhibitor, for the treatment, in combination with methylprednisolone, of patients with metastatic CRPC.7,8
The original formulation of abiraterone acetate (Zytiga) was initially FDA approved in 2011, in combination with prednisone, for the treatment of patients with metastatic CRPC who received previous chemotherapy; in 2012, the indication was expanded to include patients with metastatic CRPC, and in February 2018, abiraterone acetate was approved for the treatment of patients with high-risk metastatic castration-sensitive prostate cancer.9
This novel formulation of abiraterone acetate has a small, fine-particle size designed to increase dissolution rates and improve oral bioavailability while decreasing the effects of food on treatment efficacy.7,10 The FDA approval of the fine-particle abiraterone acetate formulation was based on the STAAR clinical trial, which demonstrated the drug’s therapeutic equivalence to the original abiraterone acetate formulation in patients with metastatic CRPC.7,10 In addition, the absorption of the 500-mg dose of the fine-particle formulation was similar to that of the 1000-mg dose of the original abiraterone acetate formulation.7,10
Mechanism of Action
CYP17 is an enzyme expressed in prostatic, testicular, and adrenal tumor tissues that is necessary for androgen biosynthesis. Abiraterone acetate is a CYP17 inhibitor and is converted to abiraterone, an androgen biosynthesis inhibitor that inhibits CYP17.8 Through the inhibition of CYP17, abiraterone blocks extragonadal and testicular androgen synthesis, thereby decreasing serum testosterone levels.10
Dosing and Administration
Abiraterone acetate is available as a 125-mg tablet.8 The recommended dosage is 500 mg (four 125-mg tablets) administered orally once daily, in combination with oral methylprednisolone 4 mg twice daily. The fine-particle formulation of abiraterone acetate tablets may have different dosing and food effects from other abiraterone acetate formulations. Patients who receive abiraterone acetate should also receive a gonadotropin-releasing hormone analog concurrently or should have had bilateral orchiectomy.8
Abiraterone acetate tablets can be taken with or without food, and should be swallowed whole with water and should not be chewed or crushed.8
Pivotal Clinical Trials
The STAAR Study
The therapeutic equivalence, pharmacokinetics, and safety of the fine-particle, 500-mg abiraterone acetate formulation plus methylprednisolone was compared with 1000-mg original formulation abiraterone acetate plus prednisone in the STAAR clinical trial, a randomized, open-label, 84-day phase 2 study in 59 men with metastatic CRPC (mean age, 75.1 years; 54.7% had a Gleason score >7).8 More than 90% of patients in each study arm achieved absolute testosterone levels of ≤1 ng/dL; average absolute testosterone levels of ≤0.1 ng/dL were demonstrated in 25% of patients in the fine-particle abiraterone acetate arm versus 17% of those in the original abiraterone acetate arm.8
At days 9 and 10, the average rounded-up least squares (LS) mean serum testosterone levels for the fine-particle formulation (1.05 ng/dL [0.04]) were comparable to those of the original formulation (1.02 ng/dL [0.03]; P = .4702) for LS mean difference. A decrease of ≥50% from baseline in prostate-specific antigen (PSA) levels was observed in >65% of patients in both arms on days 28, 56, and 84 (P = not significant, all time points). The abiraterone trough plasma levels were similar across all study visits. Furthermore, the safety profile of the fine-particle formulation was consistent with that of the original formulation.10
Study 1 and Study 2
The efficacy and safety of abiraterone acetate were evaluated in 2 randomized, placebo-controlled phase 3 studies in patients with metastatic CRPC; patients who received previous therapy with ketoconazole for prostate cancer and those with a history of adrenal gland or pituitary disorders were excluded. The concurrent use of spironolactone was not permitted.8
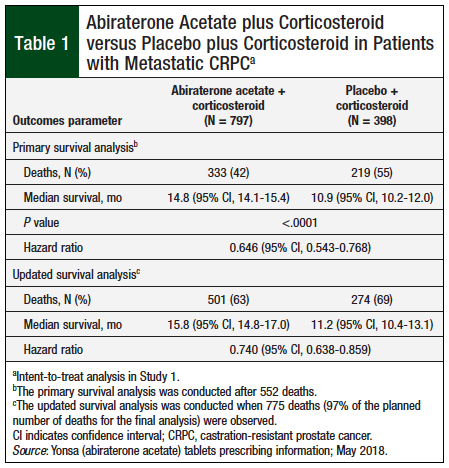
Study 1 included 1195 patients (median age, 69 years) who were randomized in a 2:1 ratio to abiraterone acetate once daily, at a dose equivalent to 500 mg of the fine-particle abiraterone acetate formulation, in combination with a different oral corticosteroid twice daily, or placebo plus oral corticosteroid twice daily. Overall, 90% of patients had bone metastases and 30% had visceral involvement.8 Patients who received abiraterone acetate demonstrated a significant (P <.0001) 3.9-month longer median survival in the primary analysis and a 4.6-month longer median survival in the updated survival analysis compared with placebo (Table 1).8
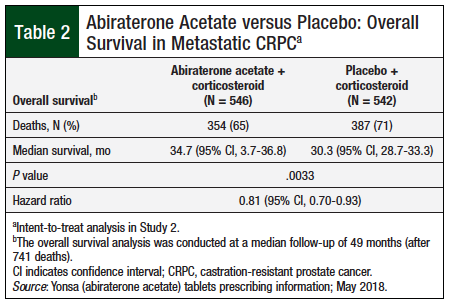
Study 2 included 1088 patients (median age, 70 years) who were randomized in a 1:1 ratio to abiraterone acetate at a dose equivalent to 500 mg of the fine-particle oral abiraterone acetate formulation once daily plus oral corticosteroid twice daily or placebo once daily plus oral corticosteroid twice daily. Patients in the abiraterone acetate group had a significant (P = .0033) 4.4-month longer median survival versus patients receiving placebo (Table 2).8
In addition, significantly fewer patients who received abiraterone acetate had radiographic progression (28%) than patients who received placebo (46%).8
Adverse Reactions
The most common (≥10%) adverse reactions reported in 2 clinical trials that occurred more often (>2%) with abiraterone acetate than with placebo were fatigue, joint swelling or discomfort, edema, hot flush, diarrhea, vomiting, cough, hypertension, dyspnea, urinary tract infection, and contusion.8
The most common (>20%) laboratory abnormalities associated with abiraterone acetate were anemia, elevated alkaline phosphatase levels, hypertriglyceridemia, lymphopenia, hypercholesterolemia, hyperglycemia, elevated aspartate aminotransferase levels, hypophosphatemia, elevated alanine aminotransferase levels, and hypokalemia.8
Based on Study 1 and Study 2, cardiac failure was more common in patients who received abiraterone acetate than in patients who received placebo (2.1% vs 0.7%, respectively). The grade 3 or 4 cardiac failure rate was 1.6% with abiraterone acetate and led to 5 treatment discontinuations and 2 deaths; the grade 3 or 4 cardiac failure rate was 0.2% with placebo, with no treatment discontinuations and 1 death from cardiac failure.8
The majority of arrhythmias in Study 1 and Study 2 were grade 1 or 2; in the abiraterone acetate groups, 1 arrhythmia-associated death and 1 sudden death occurred versus no deaths in the placebo groups. Cardiorespiratory arrest resulted in 7 (0.5%) deaths in the abiraterone acetate groups and in 3 (0.3%) deaths in the placebo groups. Death from myocardial ischemia or myocardial infarction occurred in 3 patients who received placebo and in 2 patients who received abiraterone acetate.8
Contraindications
Abiraterone acetate is not approved for use in women and is contraindicated during pregnancy.8
Drug Interactions
The concomitant use of abiraterone acetate with strong CYP3A4 inducers (ie, rifampin) should be avoided. If a strong inducer must be co-administered, the dosing frequency of abiraterone acetate should be increased.8
The co-administration of abiraterone acetate with CYP2D6 substrate drugs that have a narrow therapeutic index (ie, thioridazine) should be avoided. If an alternative treatment cannot be used, a dose reduction of the CYP2D6 substrate should be considered.8
Use in Specific Populations
Abiraterone acetate may impair reproductive function and fertility in males of reproductive potential. Males with female partners of reproductive potential should use effective contraception during treatment with abiraterone acetate and for at least 3 weeks after the final dose.8
No overall differences in the safety or effectiveness of abiraterone acetate were observed between older and younger patients.8
Abiraterone acetate should not be used in patients with baseline severe hepatic impairment (Child-Pugh Class C). In patients with mild hepatic impairment, no dose adjustment is required. In patients with moderate hepatic impairment, the recommended dose of abiraterone acetate should be reduced to 125 mg once daily.8
Warnings and Precautions
Abiraterone acetate may cause hypertension, hypokalemia, and fluid retention because of increased mineralocorticoid levels resulting from CYP17 inhibition. Patients with cardiovascular disease should be closely monitored. Before treatment with abiraterone acetate, hypertension should be controlled and hypokalemia should be corrected. Blood pressure, serum potassium levels, and symptoms of fluid retention should be monitored at least monthly.8
Patients should be monitored for adrenocortical insufficiency, particularly if the corticosteroid dose was reduced or corticosteroid treatment was withdrawn, or if the patient experiences unusual stress. An increased corticosteroid dose may be required before, during, and after stressful situations. Severe hepatic toxicity, including fatal events, can occur. Liver function should be monitored, and if necessary, abiraterone acetate should be modified, interrupted, or discontinued.8
Conclusion
The FDA approval of a novel, fine-particle formulation of abiraterone acetate provides a new oral treatment option, in combination with methylprednisolone, for patients with metastatic CRPC. In clinical trials, the fine-particle abiraterone acetate formulation demonstrated therapeutic equivalence to the original formulation, with similar reductions in PSA of ≥50%. The 500-mg dose of the fine-particle formulation was shown to have similar absorption to the 1000-mg dose of the original abiraterone acetate formulation.
References
- National Cancer Institute. SEER cancer stat facts: prostate cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed January 9, 2019.
- National Cancer Institute. Financial burden of cancer care. Updated February 2018. In: National Cancer Institute. Cancer trends progress report. February 2018. https://progressreport.cancer.gov/after/economic_burden. Accessed January 10, 2019.
- Mayo Clinic. Prostate cancer. March 9, 2018. www.mayoclinic.org/diseases-conditions/prostate-cancer/diagnosis-treatment/drc-20353093. Accessed January 11, 2019.
- American Society of Clinical Oncology. Treatment of metastatic castration-resistant prostate cancer. Cancer.Net. September 8, 2014. www.cancer.net/research-and-advocacy/asco-care-and-treatment-recommendations-patients/treatment-metastatic-castration-resistant-prostate-cancer. Accessed January 11, 2019.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Prostate Cancer. Version 4.2018. August 15, 2018. www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed January 11, 2019.
- Scher HI, Halabi S, Tannock I, et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol. 2008;26:1148-1159.
- Inman S. FDA approves new abiraterone acetate formulation for mCRPC. May 23, 2018. OncLive. www.onclive.com/web-exclusives/fda-approves-new-abiraterone-acetate-formulation-for-mcrpc. Accessed January 9, 2019.
- Yonsa (abiraterone acetate) tablets [prescribing information]. Cranbury, NJ: Sun Pharma Global; May 2018.
- US Food and Drug Administration. FDA approves abiraterone acetate in combination with prednisone for high-risk metastatic castration-sensitive prostate cancer. February 7, 2018. www.fda.gov/drugs/informationondrugs/approveddrugs/ucm596015.htm. Accessed January 11, 2019.
- Stein CA, Levin R, Given R, et al. Randomized phase 2 therapeutic equivalence study of abiraterone acetate fine particle formulation vs. originator abiraterone acetate in patients with metastatic castration-resistant prostate cancer: the STAAR study. Urol Oncol. 2018;36:81.e9-81.e16.